L- vs. D-Amino Acids
 By: by Amino Science
By: by Amino Science
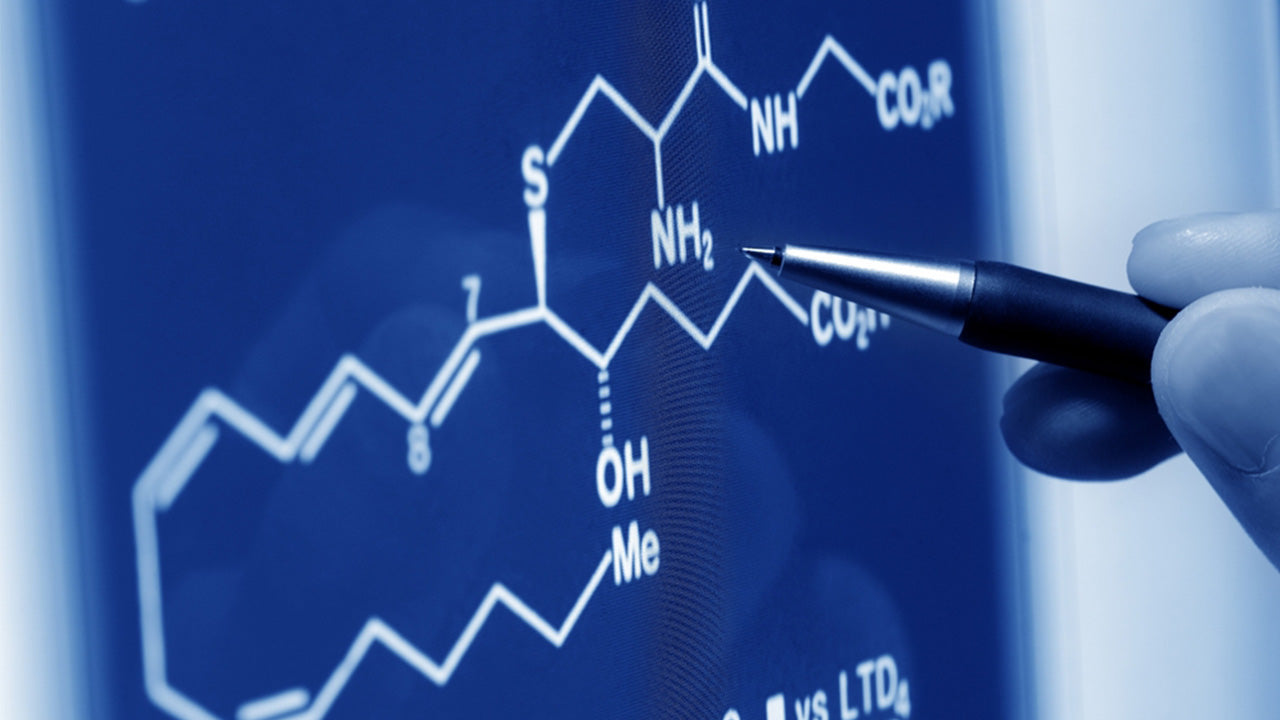
Of the 20 common amino acids in the human body that build our proteins, each of them (except for glycine) occur in two isomeric forms: L-forms and D-forms. This means that the same components of the molecules can be arranged in two different orders, a tad like how a palindrome is the same word backwards as forwards (like "radar" and "kayak"). Actually with L- vs. D-amino acids, it's more like the playful term for a word that backwards becomes a different word, a "Semordnilap" (which is "palindromes" spelled backwards). As Oprah Winfrey named her company Harpo Studios, or the town of Retsof, New York, was named for salt mine owner William Foster. Wolf vs. flow, room vs. moor, star vs. rats: the same letters, arranged exactly backwards, that then come to hold a whole new meaning. That is the basic difference between L- and D-form amino acids. The rest of this article will provide more detail and context.
What Do the L and the D Stand For?
It's easy to assume that if something is called L-form, that must mean it's in the form of an L, like a sectional sofa, or that D-form would refer to a half-moon shape, like a semi-circular booth in a restaurant, but that's not what those letters stand for here. In this context, the L and the D are referring to the order the side chain structures attach to an amino acid's central carbon atom (also known as the chiral carbon or alpha carbon). Those groups are: a single hydrogen atom (H); a carboxyl group (or COOH group); an amine group (or NH2 group); and the distinguishing R group which mostly differentiates one amino acid from another.
Picture a pair of pinwheels with four color petals (the four functional groups) attached to the centers (the alpha carbon or chiral molecules). The L-form's colors follow the alphabet and go blue, green, red, yellow if your eyes read it like a clock; your eyes read clockwise, while the wheel spins counterclockwise. The D-form goes the opposite way: to read it in the order of blue, green, red, then yellow, D-form will have to spin clockwise, while you read it backwards, eyes traveling in the counterclockwise direction.
"L" Stands for "Levorotation"
In order for your eyes to read the amino acid correctly (in alphabetical color order) the molecule itself must rotate counterclockwise, or to the left. Levorotation refers specifically to that counterclockwise rotation.
"D" Stands for "Dextrorotation"
In order for your eyes to read the amino acid in alphabetical color order, the molecule must spin clockwise, or to the right. The prefix of dextrorotation comes from the Latin, dextro, meaning "to the right." Your right hand is your dexterous one, and the left hand in the Latin would be your sinister one (many apologies to the left-handed among us; like right-handed writing desks and scissors weren't enough, you are also designated sinister as well).
How to Remember Which Is Which
If you know you're not going to remember the Latin, just remember this:
- L stands for left-moving: The molecule rotates left so you can read its structure forward, towards the right.
- D stands for your dextrous right hand: The molecule spins right so that you're reading its structure counterclockwise, backwards towards the left.
L- vs. D-Amino Acids: What Do They Do?
This section will attempt to clarify the properties of L- and D-form amino acids, what makes them different, and what they have in common.

L-Amino Acids
L-amino acids are the molecules used to produce proteins in the human body and are divided into nonessential amino acids and essential amino acids (which we must get from consuming our food). In fact, L-amino acids occur in all proteins made by animals (including humans), plants, bacteria, and fungi. They serve as both hormones and enzymes, regulating the functions in the body. L-amino acids are also the ones that can be produced by lightning reactions, possibly the origin of the organic compounds of life on Earth, and the building blocks of our proteins.
D-Amino Acids
The D-forms of amino acids are mirror images of the L-form amino acids. D-amino acids are not incorporated into proteins; however, D-serine acts as a neurotransmitter in the brain. Some D-amino acids can be found in bacterial cell walls, but again, not in bacterial proteins.
The Similarity
They are each one of the two forms that amino acids can take in nature. Normal chemical synthesis of amino acids always creates a racemic mixture, which is a mixture of equal parts L- and D-amino acids. They are mirror images of one another, and each contain a central carbon atom, a hydrogen atom, a carboxylic acid group, an amine group, and an R group carbon chain. They are like identical twins born to be the reverse of one another, as in this real case of twin boys who were nearly conjoined, but instead were born with their livers, hearts, and spleens on opposite corresponding sides of their bodies. That means one twin has a condition called dextrocardia, where his heart points towards the dextrous or right side of his body, instead of the left side. They are identical, but with opposite features, as in one of the boys is right-handed, and the other, left-handed. (Interestingly enough, there is a chance that as many as a quarter of all identical twins could be mirror twins.)
The Difference
The main difference between these two formations is the location of the amine group in their structure, which designates how the amino acid will be used. L-amino acids are used in protein synthesis, while D-amino acids are not. L-aminos rotate counterclockwise or left in a process known as levorotation, while D-amino acids rotate clockwise to the right, in what's known as dextrorotation. Usage-wise, L-amino acids are used to produce proteins, making them vitally important to our healthy functioning, while D-amino acids are found instead in the cell walls of bacteria.
Conclusion: L vs. D
Hopefully the explanation and comparison of the L- and D-form amino acids has enlightened the subject and shown you their similarities and their differences: less like dexter vs. sinister, and more like brother-to-brother twins.
Now that you know the science, how about you explore the benefits? Check out our premium amino acid supplements here.

Up to 25% off Amino
Shop NowTAGS: knowledge
Join the Community
Comments (0)
Most Craveable Recipes



 833-264-6620
833-264-6620



















